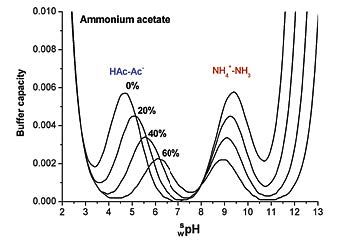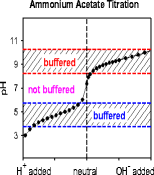
Addressing a Common Misconception: Ammonium Acetate as Neutral pH “Buffer” for Native Electrospray Mass Spectrometry | SpringerLink

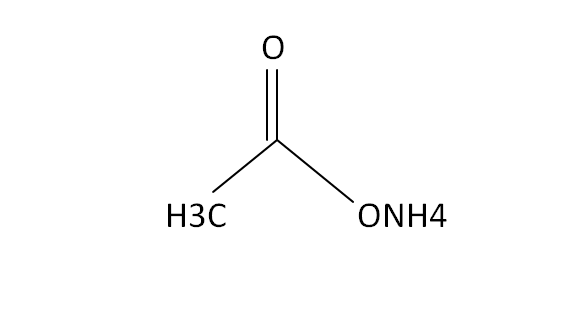
Ammonium acetate (C<sub>2</sub>H<sub>7</sub>NO<sub>2</sub>) - Structure, properties , Production, Uses and FAQs of Ammonium acetate.

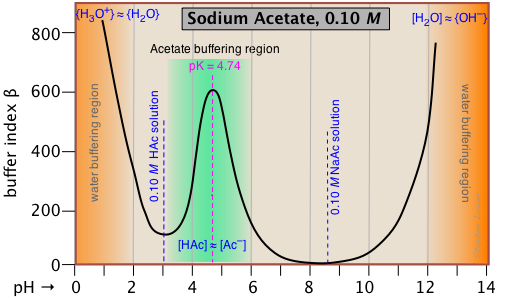
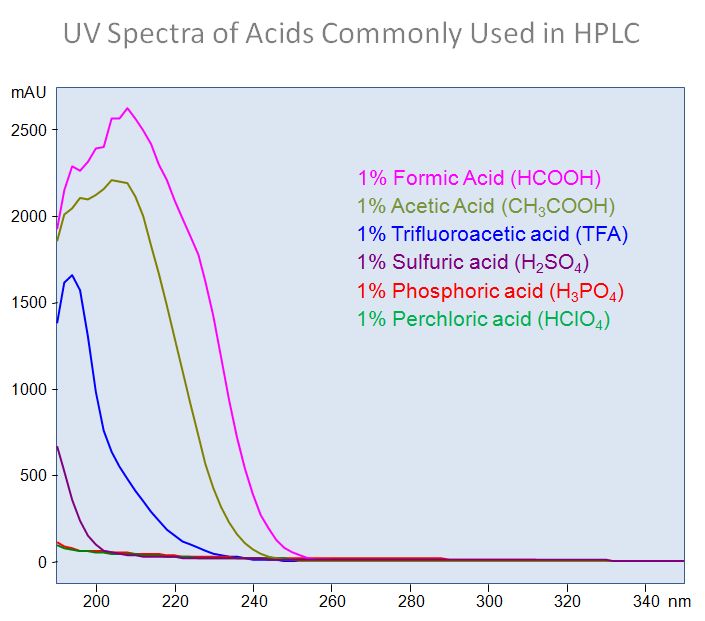
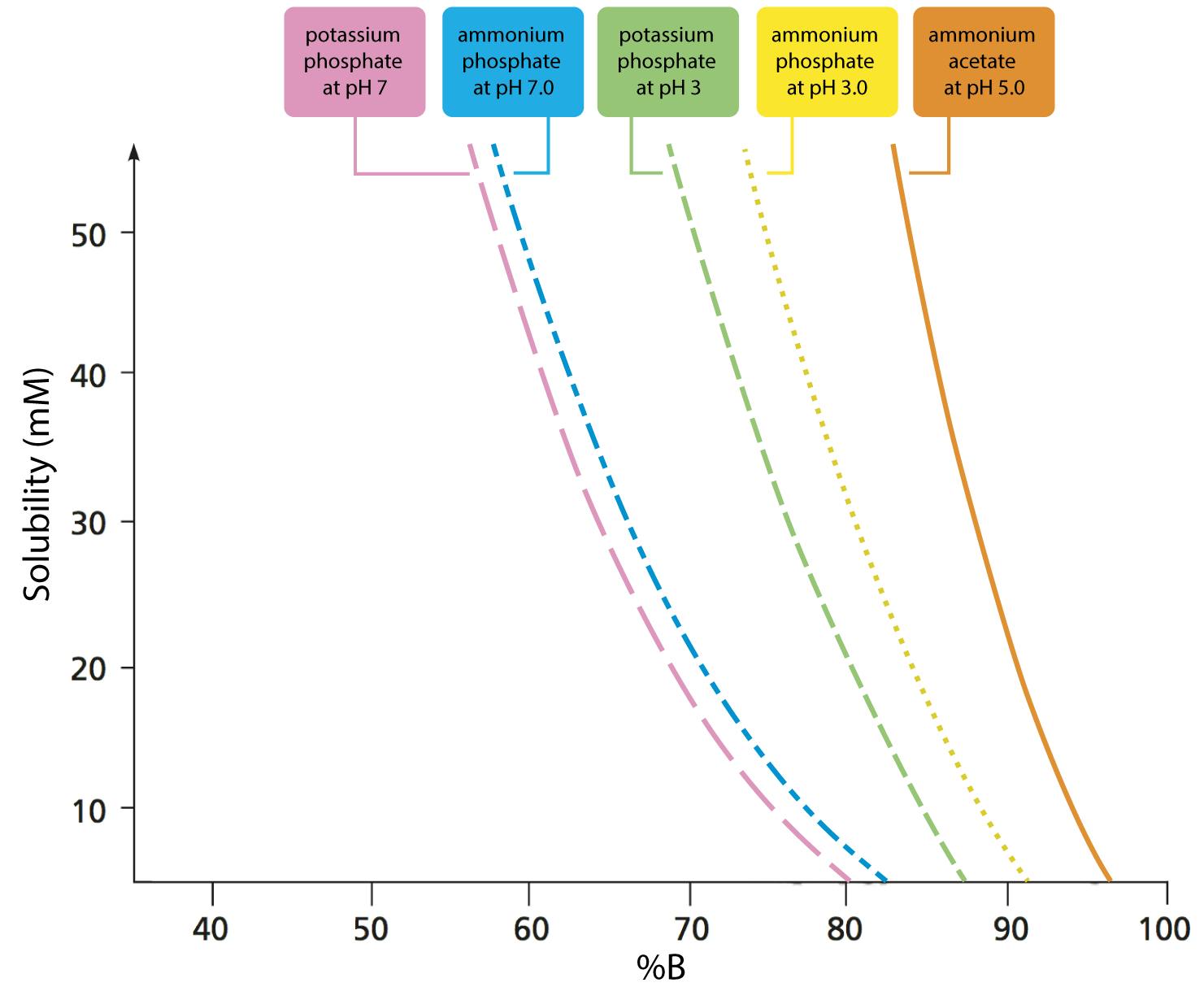
Breaking old habits: Moving away from commonly used buffers in pharmaceuticals - European Pharmaceutical Review

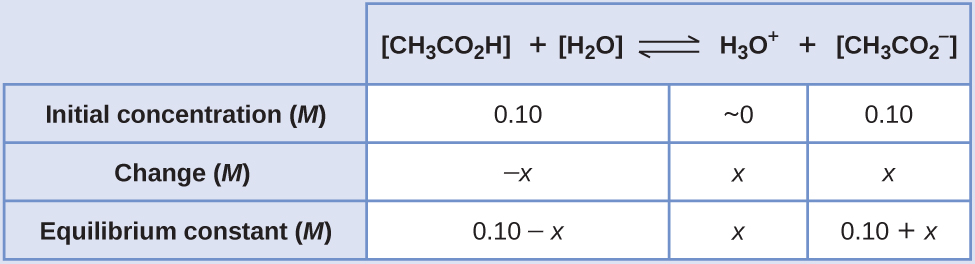
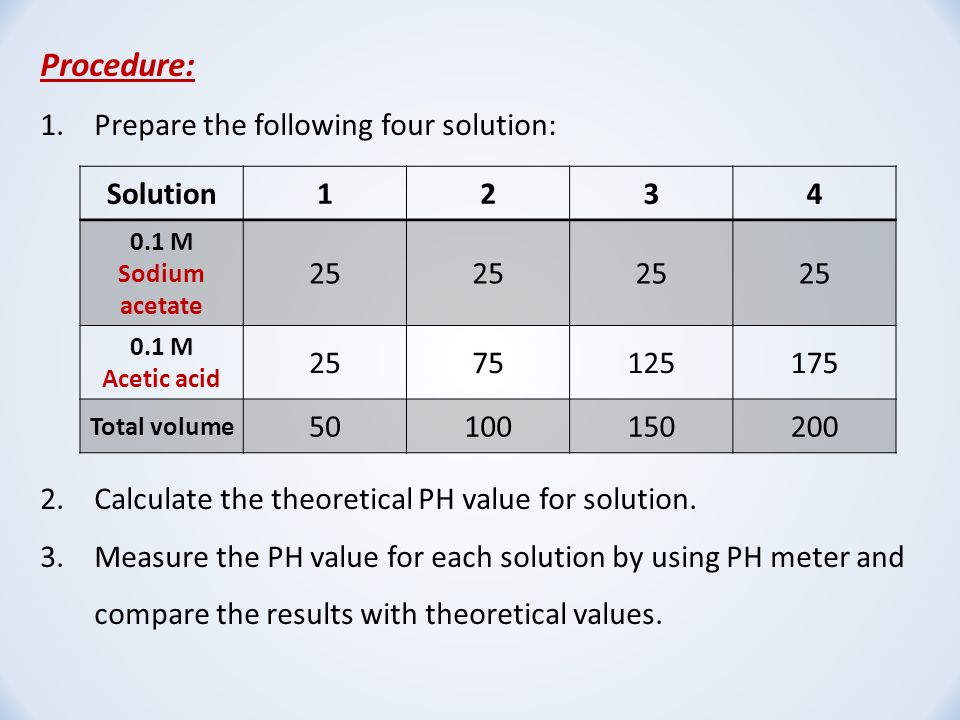
Calculate pH of 100 ml buffer solution containing 0.1 g acetic acid and 0.2 g sodium actetate. | pH calculations and more in fundamentals of pharmaceutics.




![Potassium Acetate [CH3COOK] Molecular Weight Calculation - Laboratory Notes Potassium Acetate [CH3COOK] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/potassium-acetate-molecular-weight-calculation.jpg)
![Sodium Acetate [CH3COONa] Molecular Weight Calculation - Laboratory Notes Sodium Acetate [CH3COONa] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/sodium-acetate-molecular-weight-calculation.jpg)